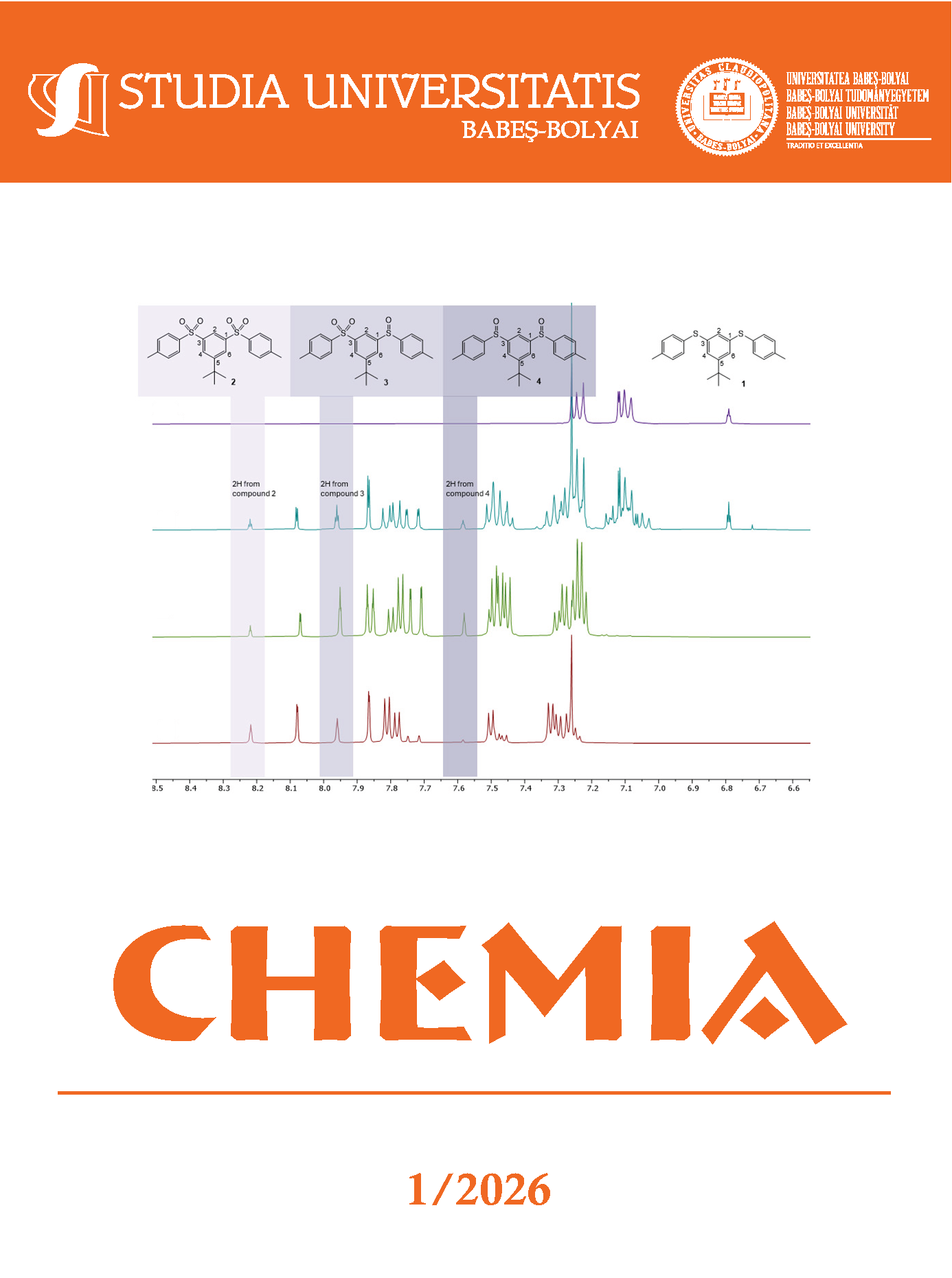
CATALYTIC SULFOXIDATION REACTIONS OF 1,3-BIS(PARA-TOLYL SULFIDE)-5-TERT-BUTYLBENZENE USING DECAVANADATE DERIVATIVES
Dedicated to Professor Luminița Silaghi-Dumitrescu on the Occasion of Her 75th Anniversary
DOI:
https://doi.org/10.24193/subbchem.2026.1.01Keywords:
catalytic sulfoxidation, bis-thioether, sulfide oxidation, green chemistryAbstract
Catalytic sulfoxidation of 1,3-bis(para-tolyl sulfide)-5-tert-butylbenzene in order to obtain sulfones and sulfoxides, was carried out using three decavanadate (V10O28)6- containing catalysts. Reaction conditions were varied to evaluate different parameters of the process; the tested procedures followed the guidelines of green chemistry. To align with these principles, reaction parameters were optimized at room temperature using environmentally benign solvents, including methanol, ethanol, iso-propanol, water, and ethylene glycol, as well as their mixtures with toluene. In this study we present a fast and environmentally friendly procedure to obtain sulfonyl and sulfinyl containing compounds through sulfoxidation of bis-thioether 1,3-bis(para-tolyl sulfide)-5-tert-butylbenzene.
References
1. M. Wang, X. Jiang, ACS Sustainable Chem. Eng. 2022, 10(2), 671–677.
2. S. Liang, K. Hofman, M. Friedrich, J. Keller, G. Manolikakes, ChemSusChem 2021, 14, 4878 – 4902.
3. J. Priego, O. García Mancheño, S. Cabrera, J. C. Carretero, J. Org. Chem. 2002, 67(4), 1346–1353.
4. G. Sipos, E. E. Drinkel, R. Dorta, Chem. Soc. Rev. 2015, 44, 3834– 3860.
5. B. M. Trost, M. Rao, Angew. Chem., Int. Ed. 2015, 54, 5026– 5043.
6. E. A. Ilardi, E. Vitaku, J. T. Njardarson, J. Med. Chem. 2014, 57(7), 2832–2842.
7. X. Chen, H. Wu, C.-M. Park, T. H. Poole, G. Keceli, N. O. Devarie-Baez, A.W.Tsang, W. T. Lowther, L. B. Poole, S. B. King, M. Xian, C. M. Furdui, ACS Chem. Biol. 2017, 12 (8), 2201–2208.
8. H. Mutlu, E. B. Ceper, X. Li, J. Yang, W. Dong, M. M. Ozmen, P. Theato, Macromol. Rapid Commun. 2018, 1800650.
9. S. Patai, Z. Rappoport, C. J. M. Stirling, The Chemistry of Sulphones and Sulphoxides, Wiley, New York, 1988.
10. E. Wojaczynska, J. Wojaczynski, Chem. Rev., 2010, 110, 4303–4356.
11. J.-E. Bäckvall, Modern Oxidation Methods, Wiley-VCH, Weinheim, 2004.
12. E. Wojaczyńska, J. Wojaczyński, Chem. Rev. 2020, 120(10), 4578–4611.
13. X. B. Liu, Q. Rong, J. Tan, C. Chen, Y. L. Hu, Front. Chem. 2022, 9, 798603.
14. J.-P. Cao, Y.-S. Xue, N.-F. Li, J.-J. Gong, R.-K. Kang, Y. Xu, J. Am. Chem. Soc. 2019, 141(49), 19487–19497.
15. W. Al-Maksoud, S. Daniele, A.B. Sorokin, Green Chem., 2008, 10, 447–451.
16. F. Rajabi, E. Vessally, R. Luque R, L. Voskressensky, Molecular Catalysis, 2021, 515, 111931.
17. N. Deak, D.Madec, G. Nemes, Eur. J. Inorg. Chem, 2020, 2769-2790.
18. N. Deak, O. Thillaye du Boullay, S. Mallet-Ladeira, I.-T. Moraru, D. Madec, G.Nemes, Eur. J. Inorg. Chem, 2020, 3729-3737.
19. N. Deak, P. M. Petrar, S. Mallet-Ladeira, L. Silaghi-Dumitrescu, G. Nemeş, D.Madec, Chem. Eur. J., 2016, 22, 1349-1354.
20. N. Deak, O. Thillaye du Boullay, I.-T. Moraru, S. Mallet-Ladeira, D.Madec, G.Nemes, Dalton Trans., 2019, 48, 2399-2406.
21. N. Deak, R. Septelean, I.-T. Moraru, S. Mallet-Ladeira, D. Madec, G. Nemes, Studia UBB Chemia, 2018, LXIII (2), 105-115
22. N. Deak, M. Idboumlik, A. Soran, M. Lachkar, B. El Bali, G. Nemes; Stud. Univ. Babes-Bolyai, Chem., 2024, 69, 7–23.
23. M. Idboumlik, M. Kadiri, N. Hamdi, M. Driouch, A.F.I. Ngopoh, I. Lakkab, E-E. Bendeif, M. Sfaira, B. El Bali, M. Lachkar, A. Zarrouk, Mater. Chem. Phys., 2022, 287, article no. 126211.
24. M. Idboumlik, I. Lakkab, S. Erraouan, N. Hamdi, M. Lachkar, M. Dusek, V. Eigner, B. El Bali, J. Mol. Struct., 2024, 1312, article no. 138551.
25. P. T. Anastas and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, 1998.
26. A.M. Afanasenko, N. Deak, J. October, R. Sole, K. Barta, Green Chem., 2025, 27, 5947-5981.
27. D. Prat, A. Wells, J. Hayler, H. Sneddon, C.R. McElroy, S. Abou-Shehadad, P.J.Dunn, Green Chem., 2016,18, 288-296.
28. D. Prat, J. Hayler, A. Wells, Green Chem., 2014, 16, 4546-4551.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Studia Universitatis Babeș-Bolyai Chemia

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.